Sialorrhea in Cerebral Palsy
Sialorrhea in Cerebral Palsy
- Section I: Evidence Summary
- Section II: Published Evidence
- Section III: Practical Tools
- Section IV: Acknowledgments
- Feedback/
Comments
Section I: Evidence Summary Printer Friendly Version
Definition
Sialorrhea refers to drooling of saliva as a result of limitations in a person’s ability to control and swallow oral secretions. Anterior drooling is defined as saliva spilled from the mouth that is clearly visible. Posterior drooling occurs when saliva spills through the oropharynx and into the hypopharynx. In children and youth with cerebral palsy (CP), sialorrhea is usually the result of limited oromotor control as a result of muscle incoordination and sensory perception difficulties rather than excessive salivation.
Why is Sialorrhea Important?
Sialorrhea occurs in approximately 40% of children/youth with CP and can have significant medical and psychosocial impact.
Medical concerns:
- Posterior pooling can have the serious consequence of chronic aspiration resulting in recurrent infections and progressive lung disease.
- Presence of saliva on the chin leads to frequent wiping, causing skin irritation and breakdown.
Psychosocial concerns:
- Anterior drooling of saliva may result in the need for frequent clothing changes, may damage books, computers, toys and other equipment, and spray from the mouth while talking.
- Social embarrassment experienced by children/youth, their caretakers and siblings can be considerable and may lead to isolation and low self-esteem.
Target Population: Children/youth between the ages of birth and 25 years with CP who drool
Target Clinical Providers: Physicians, therapists, psychologists and nurses treating children/youth with CP who drool
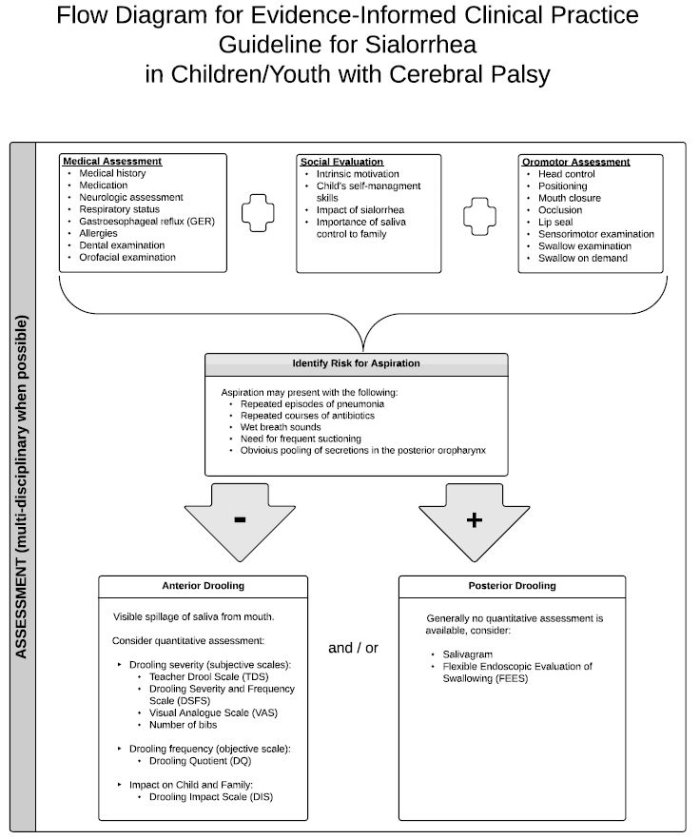
Assessment
- Discussion with providers from multiple disciplines is recommended
- Medical assessment
- Emphasis on medications, history of aspiration, respiratory status and lower airway examination, neurologic assessment (craniofacial control, posture, impact of medicines, epilepsy, developmental age equivalent), gastroesophageal reflux disease (GERD), presence of allergy, orofacial examination (dentition, oral hygiene, upper airway), hydration.
- Social evaluation
- Intrinsic motivation and child’s self-management skills, impact of sialorrhea and importance of saliva control to family
- Motor/Oromotor assessment
- Head control, positioning, mouth closure, occlusion, lip seal, sensorimotor evaluation, swallow on demand, ability to wipe own saliva
- Drooling can be assessed quantitatively with a variety of tools for severity and frequency as well as impact on the child and family [Drooling Quotient, Teacher Drooling Scale, Drooling Severity and Frequency Scale, Visual Analogue Scale (VAS), Drooling Impact Scale (DIS), number of bibs, frequency of clothing changes]. Quantification can aid in gauging response to interventions.
- Differentiation between anterior and posterior drooling is important. They may appear independently or may co-exist. Most often, clinical information such as repeated episodes of pneumonia, repeated antibiotic courses for respiratory reasons, evidence of chronic inflammatory lung disease, and significant need for suctioning are used as indicators of posterior drooling. Additional investigations to consider include salivagram and fiberoptic endoscopic evaluation of swallowing; however, they may not be necessary or appropriate.
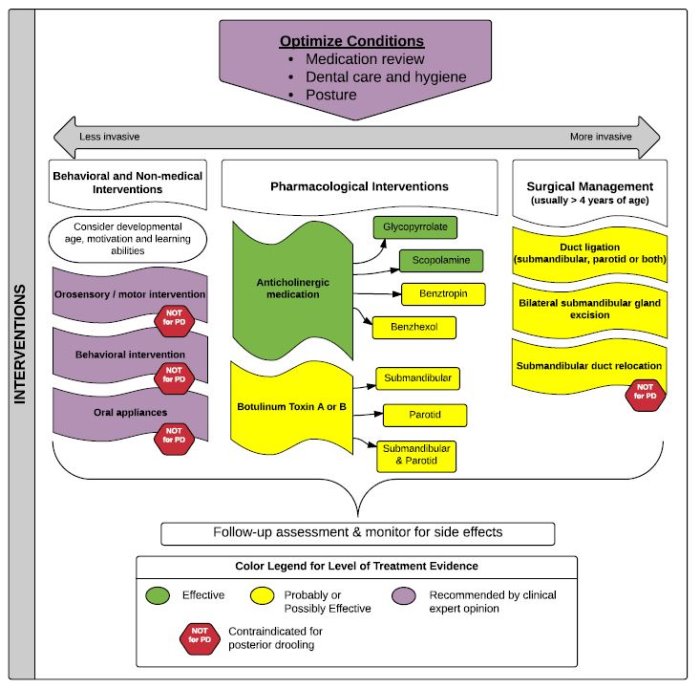
Treatment Options
A number of treatment strategies are available although there is no clear consensus as to which are safe and effective. Goals of treatment target: 1) improvement of oromotor control of secretions; 2) enhancement of a child’s ability to behaviorally manage secretions; and 3) reduction of saliva production or rerouting of salivary flow. When possible, a multidisciplinary team approach is recommended, progressing from conservative to more invasive treatments until saliva control is improved and side effects, if present, are manageable. Complete control is often not possible. Additionally, surgical intervention may not be curative. All of the strategies that follow may be appropriate for anterior drooling; oromotor and orosensory strategies, behavioral strategies, and oromotor appliances are not recommended for posterior drooling. Duct relocation is contraindicated for posterior drooling.
- Optimize conditions - Optimize positioning and medical management of factors that affect drooling. Consider whether medications being used for other conditions, such as epilepsy, are increasing drooling.
- Oromotor and orosensory strategies - Active and passive exercises as well as sensory applications are widely used by clinicians, although there is no agreement about the theoretical basis and effectiveness of these interventions. These approaches can be time consuming. No adverse effects are reported.
- Behavioral strategies - Multiple types of behavioral procedures have been shown to be effective (low level evidence). Selection and success depends on the ability of the child to comply and often requires on-going effort for maintenance of effect. No adverse effects are reported.
- Oral appliances - Compliance can be challenging and nose breathing must be possible for the child wearing the appliance. Children with seizure disorders may be at risk for oral injury. There is some low level evidence that oral appliances may be effective.
- Anti-cholinergic agents which inhibit salivary secretion - Glycopyrrolate, scopolamine (also known as hyoscine), benzhexol and benztropin are the most commonly used agents internationally. These medications, while effective, are sometimes associated with adverse side effects such as excessive thickening of secretions, urinary retention, constipation, headache, blurred vision and behavioral disturbance.
- Intraglandular Botulinum toxin injections to the submandibular +/- parotid glands - Injections are often considered after inadequate response to anti-cholinergic treatment. They can be effective but need to be repeated regularly, often at 6 month intervals, and responsiveness may diminish over time. Botulinum toxin is most often injected using ultrasound guidance for assistance. Varying doses have been reported with botulinum toxin A being the most frequently used type. Side effects include irritation at the injection site, pain, hematoma, dry mouth, thickened secretions, or problems with chewing and swallowing from diffusion to the surrounding muscles thus, increasing aspiration risk.
- Surgical intervention. Surgery is usually reserved for patients with profuse, persistent anterior drooling, continued symptoms despite maximal conservative or pharmacological treatment, and patients with posterior drooling who have chronic aspiration and/or recurrent respiratory infections. Surgical procedures may include duct ligation or rerouting, sublingual or submandibular gland excision, and varying combinations of these procedures. Success and caregiver satisfaction are variable. Duct recanalization can occur. Side effects are usually minimal but include xerostomia and wound infection.
Longitudinal Management
Whether or not an intervention is utilized, the psychosocial and medical effects of drooling must be monitored longitudinally. If an intervention is pursued, regular systematic monitoring of the child and caretaker for indications of efficacy and potential side effects is imperative.
Algorithm
Section II: Published Evidence
A Cochrane report was available on the subject of sialorrhea in children with cerebral palsy and framed the core of this summary and algorithm. Additionally, a broad literature search was conducted to complement the report. This included review of lower level evidence studies which the team felt might further inform the range of choices clinicians confront in the treatment of sialorrhea. The results are being submitted for publication as a review article which will be posted after publication.
 |
Interventions for drooling in children with cerebral palsy. Walshe M, Smith M, Pennington L. Cochrane Database of Systematic Reviews 2012, Issue 11. Art. No.: CD008624. DOI: 10.1002/14651858.CD008624.pub3 |
Section III: Practical Tools
Plain Language Summary
Print Material
 |
Saliva Control in Children Royal Children’s Hospital Melbourne |
Mobile App
 |
Swallow Prompt Note: There may be a small fee associated with this mobile app. |
Measurement Tools
 |
Drooling Quotient Instructions (5-minute version) |
 |
Drooling Impact Scale |
Section IV: Acknowledgments
| Name | Affiliation(s) | Location | Specialty Expertise |
|---|---|---|---|
| Corinne Delsing | Radboud University Medical Centre | Nijmegen, The Netherlands | Otorhinolaryngology |
| Laurie J. Glader, MD | Boston Children's Hospital Harvard Medical School |
Boston, Massachusetts, USA | Pediatrician, Complex Care Medicine |
| Amy Hughes | Boston Children's Hospital Harvard Medical School |
Boston, Massachusetts, USA | Pediatric Otolaryngology |
| Jeremy Parr, MB ChB, MRCPCH, MD | Institute of Neuroscience, Newcastle University | Newcastle upon Tyne, UK | Clinical Senior Lecturer and Consultant, Paediatric Neurodisability |
| Lindsay Pennington, PhD | Institute of Health and Society, Newcastle University | Newcastle upon Tyne, UK | Speech and Language Therapy |
| Dinah S. Reddihough, MD | Royal Children's Hospital Murdoch Children's Research Institute The University of Melbourne |
Parkville, Victoria, Australia | Developmental Medicine |
| Karen Van Hulst, MSc | Radboud University Medical Centre | Nijmegen, The Netherlands | Speech and Language Therapy/ Clinical Epidemiology |
| Jan JW Van Der Burg, PhD | St. Maartenskliniek & Radboud University | Nijmegen, The Netherlands | Psychologist |

Feedback/Comments
The American Academy for Cerebral Palsy and Developmental Medicine has developed care pathways to assist the busy clinician. Please submit any advice or constructive feedback to make this pathway more useful.
NOTE: Feedback will be directed to the AACPDM Care Pathway Taskforce to review and consider on a queue 6-month basis.